Can gamma-proof data stem the tide of aseptic manufacturing deficiencies?
In news that’s starting to sound like a broken vinyl record, instances of voluntary recall and FDA warnings continue to plague the aseptic manufacturing industry. To wit:
Baxter issued a voluntary recall for more than 427,000 units of sodium chloride injection and 54,528 containers of dextrose injection, citing “a lack of assurance of sterility” as the driving mechanism. (Read more on FiercePharma).
The FDA cited Tubilux for “deficiencies that include improper equipment use, insufficient laboratory controls, and problems with the company’s sterility assurance program.” (Read more on PharmTech.com).
And most recently, Rugby Laboratories just issued a major voluntarily recall for Diocto Liquid and Diocto Syrup. (Read more on Pharmaceutical Processing).
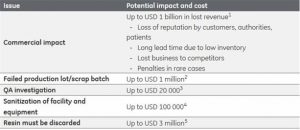
Even as the global market for environmental monitoring is estimated to reach $19.56 Billion by 2021, factors such as high costs of current proposed solutions, complicated implementation procedures, and high export barriers across emerging countries are restraining market growth.
How can gamma-survivable digital intelligence help? In an aseptic manufacturing environment, it transforms the very assets already in place into smart aids that keep better track of moment-by-moment conditions and process controls, to produce a more complete and verifiable record of sterility assurance.
The assets we’re talking about are those that monitor airborne particulates, active viable air, passive viable air and equipment surfaces, and facility personnel themselves. Whenever a drug or biologic goes through a given process or stage of production, these components gather digital records and time-stamped details about the manufacturing procedure, location or condition of the environment, which of course includes chain-of custody and information needed for regulatory compliance. These assets become embedded with a literal digital thread, to help downstream operators collect, manage, and report every stage of production including initial sterilization. The data then feeds the manufacturing clinical laboratory database and, quite simply, personnel are put in position to perform their jobs better. Operators, laboratory technicians, managers — even executives — can digitally access and sync component data, and call up production or sterility details about any individual unit at any time, even after a batch has been released to the market.
To learn about Tego’s gamma-proof intelligent solutions for pharmaceutical manufacturing, please visit this page.
To schedule a demo and see how Tego can improve your aseptic manufacturing processes, contact us here.